HAVE ANY QUESTIONS? CLICK HERE
Resorbable Collagen Membrane – Guided bone Regeneration
EZ Cure™ Membrane is a highly purified porcine collagen (type I and III) resorbable membrane.
Phylogenetically, porcine collagen is the closest to human collagen. Porcine tissue is viewed as the material of choice in many medical xenograft procedures.8
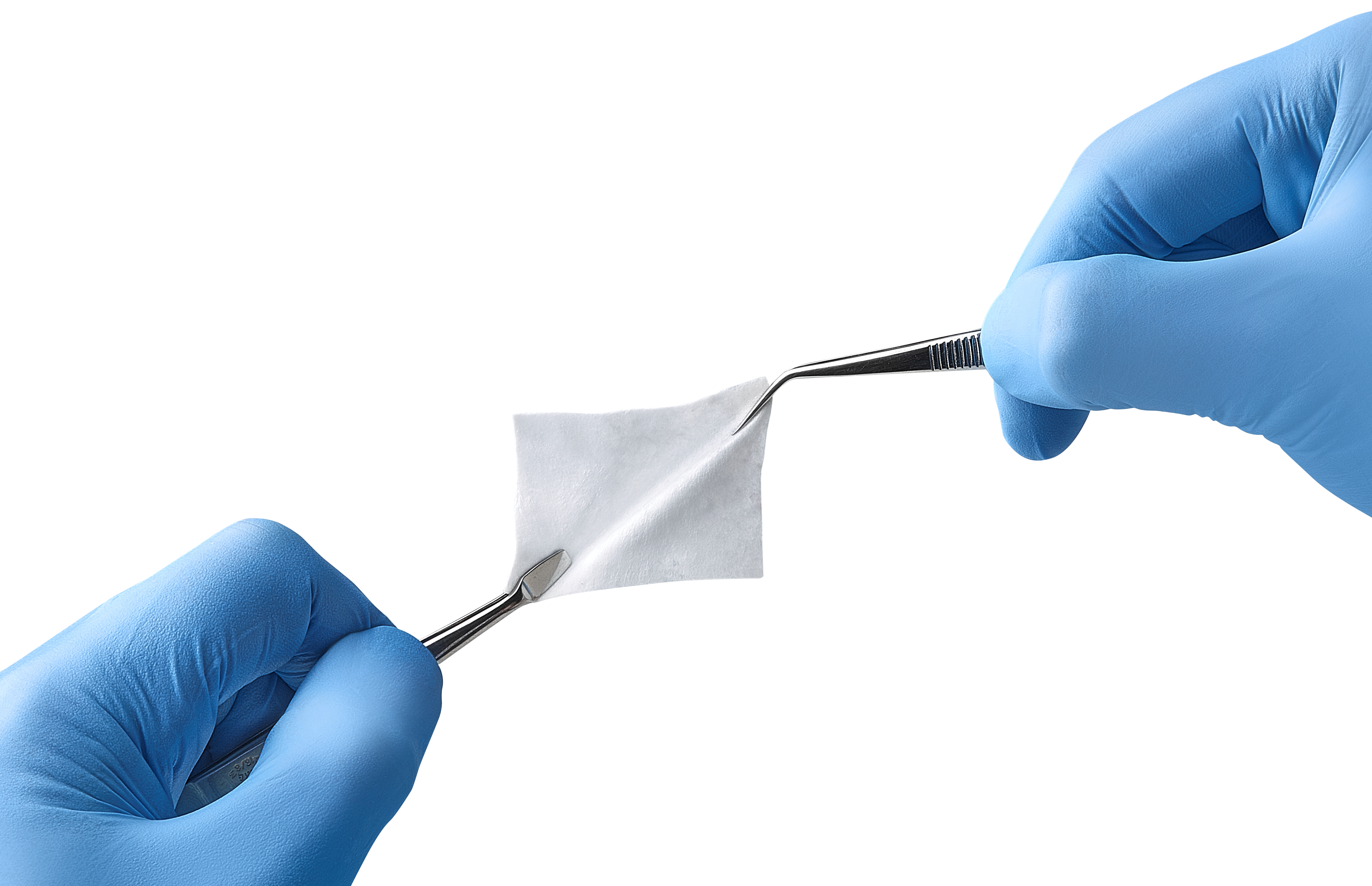
EZ Cure™ Resorbable Membrane offers excellent handling properties.*
Its adaptability to different bone geometries makes the surgical procedure easier and effective.*
Barrier function for at least 3 months
EZ Cure™ Membrane is a cross-linked collagen membrane that combines controled resorption time (barrier function) and flexibility*.
EZ Cure™ Membrane ensures a barrier function for a minimum of 3 months* in order to prevent connective or epithelial cells from colonizing the surgical site during the healing period.
- Flexible and adaptable to varying bone topographies
- Great tissue adherence
- Cell occlusive
- Easy to cut, to shape and to place
- Strong, can be sutured